1.800.858.7378npic@oregonstate.edu
Call, email, or chat Mon-Fri
A to Z
Medical Case Profiles
Medical Cases and Topics for Health Care Providers are based upon inquiries received by NPIC, as well as relevant publications in the scientific literature. They are intended to educate health care providers about pesticide toxicology.
Medical Case Profiles - Biomarkers of Exposure: Organophosphates
Scenario:
A healthy adult female visits her physician for an annual examination. She has heard reports on television about recent studies that have measured pesticide residues in urine samples taken from the United States population. She would like to know more about testing for exposure to organophosphates, as she has used insecticides containing these active ingredients in the past at her residence. She is seeking information from the physician about testing for exposure to this group of insecticides, and how to interpret the results.
Discussion:
Biomarkers of exposure are important in toxicology, because they are an indicator of internal dose, or the amount of chemical exposure that has resulted in absorption into the body. Significant advances have been made in the development of analytical methods which can detect and/or quantify the presence of many natural or synthetic toxins or their breakdown products (metabolites) in a biological matrix (such as blood or urine). The ability to accurately measure biomarkers of exposure depends upon an adequate understanding of the chemistry and toxicology of the substance under consideration.
Organophosphate insecticides share a common mechanism of toxicity, through inhibitory effects on cholinesterase enzymes in the nervous system.1 In addition, all organophosphates share some common chemical properties. Organophosphates contain a central phosphorus atom with a double bond to either sulfur or oxygen, R1 and R2 groups that are either ethyl or methyl in structure, and a leaving group which is specific to the individual organophosphate. The general structure of organophosphates, and a specific example (chlorpyrifos) appear below:

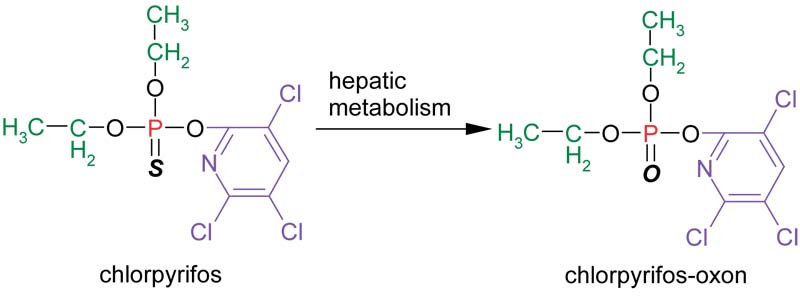
Organophosphates undergo chemical changes in the environment as well as in the human body. It is necessary to understand these changes in order to measure and interpret biomarkers of exposure to organophosphates. One chemical reaction that occurs in humans is a transformation at the double bond of the central phosphorus atom from sulfur to oxygen. This metabolic reaction takes place in the liver and results in activation of the organophosphate to a more potent inhibitor of cholinesterase enzymes. The metabolic activation from chlorpyrifos to chlorpyrifos-oxon appears below:
Another important metabolic reaction that occurs in the human body is hydrolysis of the organophosphate, yielding a dialkylphosphate and the leaving group. This reaction results in a decrease in toxicity, as the leaving group and dialkylphosphate metabolites do not inhibit cholinesterase enzymes. This metabolic detoxification reaction is shown for chlorpyrifos and chlorpyrifos-oxon below:
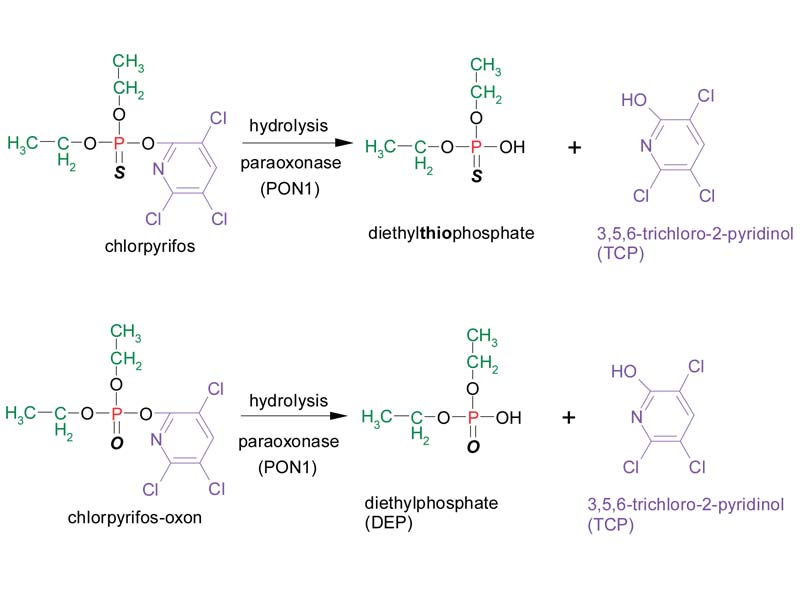
These products of hydrolysis can be measured as biomarkers of exposure to organophosphates, as the metabolites are subsequently eliminated from the body in the urine. Because the chemical structure of the leaving group is specific to the organophosphate, the detection and quantification of the leaving group is a relatively specific biomarker of exposure to the parent compound. For example, the leaving group 3,5,6-tricholoro-2-pyridinol (TCP) can be measured in the urine as a relatively specific biomarker of exposure to chlorpyrifos.
An additional factor that may affect the interpretation of leaving groups as biomarkers of exposure is the observation that certain organophosphates may undergo hydrolysis reactions in the environment. Chlorpyrifos, for example, undergoes hydrolysis in the environment to yield the leaving group 3,5,6-tricholoro-2-pyridinol (TCP) as the major degradation product.2 For this reason, in addition to reflecting exposure to the parent compound (chlorpyrifos), the level of leaving group metabolites in the urine may also be an indicator of exposure to TCP itself, if it were present in an individual's environment. This poses a challenge in interpreting the human health significance of metabolite levels, as TCP does not inhibit cholinesterase enzymes.3
The interpretation of urinary dialkylphosphates is also complicated because the hydrolysis of a specific organophosphate may yield more than one class of dialkylphosphates.4 This is apparent in the case of chlorpyrifos, where the products of hydrolysis can include both diethylphosphate (DEP) as well as diethylthiophosphate, depending upon whether the chlorpyrifos has undergone metabolic activation to chlorpyrifos-oxon in the body. For this reason it is not possible to determine which specific organophosphate an individual was exposed to based upon the detection or quantification of dialkylphosphate metabolites in the urine.
Recent studies have quantified biomarkers of exposure to organophosphates in large samples of the United States population,5 using urinary dialkylphosphate and other metabolites. While this research presents important information about exposure to organophosphate insecticides and their metabolites in the population, generally recognized guidelines for the interpretation of these biomarkers of exposure have not been established.4 The detection of urinary alkylphosphates in the urine does not necessarily indicate that they are the cause of an adverse health effect. The relationship between urinary dialkylphosphates and acetylcholinesterase enzyme activity has not been studied in the general population. More research is needed to determine the relationship between these biomarkers of exposure and health effects, as well as the relative role of dietary, residential, and occupational pathways of exposure.
While the development and interpretation of biomarkers of exposure to organophosphate insecticides is an important area of research, other biomarker methods are available to physicians in the clinical assessment of acute exposure. The activity of cholinesterase enzymes in the blood can be measured as a biomarker of effect for organophosphates. An individual with acute symptomatic overexposure to organophosphates will usually have an abnormally low level of activity of cholinesterase enzymes measured in the serum (as butyrylcholinesterase, also known as pseudocholinesterase) or in red blood cells (as RBC cholinesterase, which is more closely correlated with acetycholinesterase activity in the nervous system). Blood cholinesterase measurements have limitations in that the time course for enzyme inhibition and recovery can vary with exposure to different organophosphates. Cholinesterase activity can also be affected by inter- and intra-individual variability. However, in combination with a careful exposure history,6 their measurement may provide important additional information to the health care provider evaluating an individual with acute overexposure to organophosphate insecticides. In an asymptomatic, healthy individual without a history of recent overexposure to organophosphate insecticides (as in the current case scenario), cholinesterase testing is not likely to be of clinical value. Guidelines have been developed for physicians involved in the surveillance of workers with potential exposure to organophosphates, using serial monitoring of cholinesterase enzyme activity in the blood.
Current research is also investigating the potential that individuals with variable activity of certain enzymes important to the metabolism of organophosphates may be more or less susceptible to their effects. One important enzyme that is under investigation as a biomarker of susceptibility is paraoxonase (or PON1), which is responsible for the hydrolysis and deactivation of organophosphates.7 As polymorphisms in the PON1 gene exist in humans, and such polymorphisms may affect the efficiency with which an individual will metabolize organophosphates, biomarkers of genetic susceptibility may prove to be important in assessing an individual's risk of adverse effects from excessive exposure. At this time, however, such biomarkers of susceptibility have not been adequately studied and validated to incorporate into clinical practice.
References:
- Organophosphate Insecticides. In: Reigart JR, Roberts JR, eds. Recognition and Management of Pesticide Poisonings (Fifth Edition), 1999:34-48.
- U.S.Environmental Protection Agency, Office of Pesticide Programs. Interim Reregistration Eligibility Decision for Chlorpyrifos: Section IIIB: Environmental Risk Assessment. Pages 43-60. February, 2002.
- U.S.Environmental Protection Agency, Office of Pesticide Programs. Interim Reregistration Eligibility Decision for Chlorpyrifos: Section IIIA1a, Pages 11-12. February, 2002.
- Centers for Disease Control, National Center for Environmental Health, Atlanta, Georgia. Organophosphorus Insecticides: Dialkyl Phosphate Metabolites. Pages 117-133. 2009. In: Fourth National Report on Human Exposure to Environmental Chemicals.
- Centers for Disease Control, National Center for Environmental Health, Atlanta, Georgia. Fourth National Report on Human Exposure to Environmental Chemicals. Page 3. 2009.
- Environmental and Occupational History. In: Reigart JR, Roberts JR, eds. Recognition and Management of Pesticide Poisonings (Fifth Edition), 1999:17-32.
- Costa LG, Cole TB, Furlong CE. Polymorphisms of paraoxonase (PON1) and their significance in clinical toxicology of organophosphates. J Toxicol Clin Toxicol. 2003;41(1):37-45.

